Abstract:
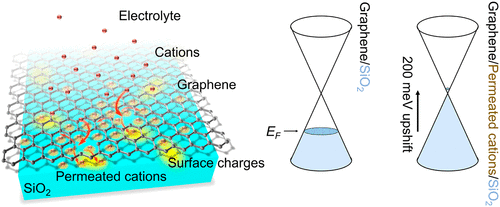
Due to its outstanding electrical properties and chemical stability, graphene finds widespread use in various electrochemical applications. Although the presence of electrolytes strongly affects its electrical conductivity, the underlying mechanism has remained elusive. Here, we employ terahertz spectroscopy as a contact-free means to investigate the impact of ubiquitous cations (Li+, Na+, K+, and Ca2+) in aqueous solution on the electronic properties of SiO2-supported graphene. We find that, without applying any external potential, cations can shift the Fermi energy of initially hole-doped graphene by ∼200 meV up to the Dirac point, thus counteracting the initial substrate-induced hole doping. Remarkably, the cation concentration and cation hydration complex size determine the kinetics and magnitude of this shift in the Fermi level. Combined with theoretical calculations, we show that the ion-induced Fermi level shift of graphene involves cationic permeation through graphene. The interfacial cations located between graphene and SiO2 electrostatically counteract the substrate-induced hole doping effect in graphene. These insights are crucial for graphene device processing and further developing graphene as an ion-sensing material.